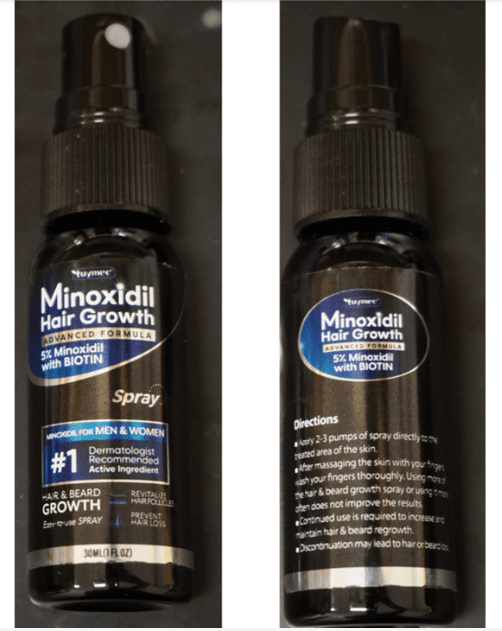
Tuymec Minoxidil Hair Growth Kit Recalled for Non-Child-Resistant Packaging (2026)
Tuymec Minoxidil Hair Growth Kit sold on Amazon by DrHealBeauty is recalled for non-child-resistant packaging. The kit contains two black spray bottles with 5% minoxidil and BIOTIN labels. Consumers should stop using the product immediately and contact DrHealBeauty for a free empty replacement bottle with a child-resistant closure.




